
Medical Writing
Medical Writing Services
At ABRS, we specialize in delivering high-quality Medical Writing Services through a Functional Service Provider (FSP) model that supports sponsors and CROs worldwide. Our methodology is based on flexibility, scalability, and regulatory compliance, ensuring research teams can accelerate timelines without compromising on accuracy or quality.
¿Why choose ABRS medical writing services?
In today’s complex clinical research environment, high-quality medical writing is essential to the success of a study. Clinical trials rely on experienced medical writers to develop clear, accurate, and regulatory-compliant documents that support study conduct, regulatory submissions, and stakeholder communication.
ABRS medical writing services ensure that critical clinical documents meet global regulatory standards and effectively support interactions with regulators, investigators, ethics committees, and sponsors throughout the trial lifecycle.
A Flexible FSP Model for Every Research Need
ABRS’s Functional Service Provider (FSP) model is designed to support the evolving medical writing needs of clinical research programs. Recognizing that documentation demands vary by study phase, therapeutic area, and regulatory pathway, we offer flexible engagement options that provide access to experienced medical writers when and where they are needed.
Our FSP approach enables seamless integration with sponsor and CRO teams, ensuring continuity, consistency, and regulatory alignment across protocols, clinical reports, and other essential study documents.
- Scalability
- Compliance
- Consistency
- Efficiency




A Flexible FSP Model for Every Research Need
Whether you need full-time dedicated writers embedded within your team, temporary support during periods of peak workload, or scalable capacity to cover multiple global studies, we adjust our services to fit seamlessly into your processes.
- FAQ
FAQ’s
A Functional Service Provider (FSP) is a clinical research outsourcing model in which sponsors engage specialized professionals to support specific functional areas of a clinical trial, such as clinical operations, regulatory affairs, data management, and pharmacovigilance.
Unlike traditional outsourcing models, the FSP approach allows sponsors to retain strategic control of their studies while strengthening internal teams with specialized expertise. At Advanced BioResearch Solutions (ABRS), the FSP model integrates experienced professionals directly into sponsor teams, helping organizations improve operational efficiency and ensure high-quality clinical trial execution.
The FSP model provides several advantages for sponsors conducting clinical trials, including greater flexibility, scalability, and access to specialized expertise.
Organizations can strengthen specific operational functions without outsourcing entire studies, allowing them to optimize resources, maintain oversight of clinical strategies, and adapt teams as project needs evolve. This model also supports stronger collaboration between sponsor teams and external experts, helping improve efficiency and regulatory compliance.
In a traditional Contract Research Organization (CRO) model, the CRO typically manages large portions or the entirety of a clinical trial. In contrast, the FSP model focuses on providing dedicated professionals who support specific functional areas within the sponsor’s organization.
This approach allows sponsors to retain strategic oversight while strengthening internal capabilities with specialized resources, offering greater flexibility, transparency, and operational alignment with sponsor processes and systems.
Our FSP model stands out through the strength of our people, the discipline of our execution, and our unwavering commitment to compliance.
At the core is our talent: highly experienced professionals who bring not only technical expertise but also a proactive, ownership-driven mindset. They integrate seamlessly into sponsor teams, enabling faster ramp-up, stronger collaboration, and consistent delivery of high-quality outcomes.
We also take a strategic approach to budget management. By aligning resources closely with project priorities, we ensure cost efficiency while maximizing operational impact—allowing sponsors to maintain control without compromising performance or timelines.
Finally, everything we do is grounded in strict adherence to global regulatory standards and industry best practices. This ensures that every activity is compliant, audit-ready, and aligned with the highest quality expectations, reducing risk and strengthening overall study integrity.
The ABRS FullSpectrum FSP model provides sponsors with access to experienced professionals across the full range of clinical research functions. Available roles include Senior Clinical Research Associates (Sr. CRAs), Clinical Oversight Managers, Regulatory Affairs specialists, Data Management professionals, Pharmacovigilance experts, and Auditors — each deployed based on the specific operational needs of the study.
What distinguishes ABRS is not simply the availability of these roles, but how they are integrated. Each professional embeds directly into the sponsor’s team, operating under sponsor systems, SOPs, and governance structures. This ensures continuity, accountability, and alignment with the sponsor’s quality standards from day one.
Whether supporting a single functional area or spanning multiple workstreams across global regions, ABRS resources are selected for both technical.
Sponsors often use the FSP model when they need to strengthen specific operational functions without fully outsourcing clinical trial management.
This model is particularly beneficial when organizations require specialized expertise, scalable FullSpectrum FSP solutions, or additional resources to support expanding clinical programs. The FSP approach enables sponsors to scale teams efficiently while maintaining control over clinical development strategies.
Our Location
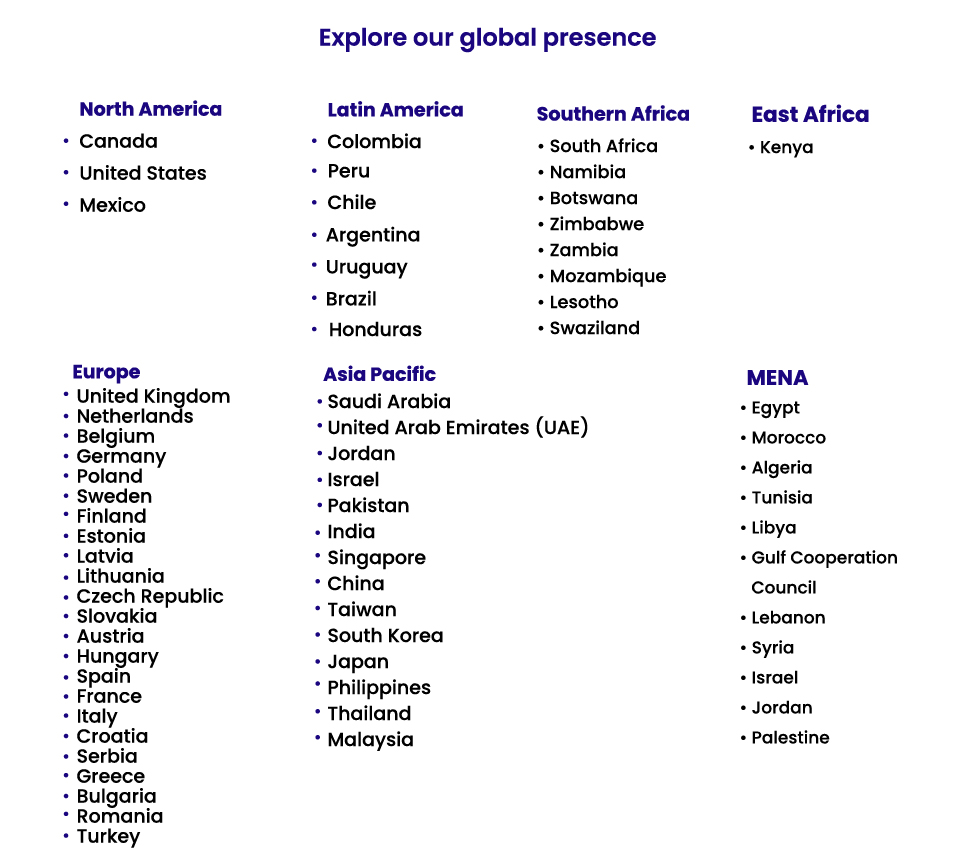
At ABRS, we currently provide coverage
North America
Latin America
East Africa
Southern Africa
Europe
Asia-Pacific
MENA

Join us as an ABRS AMBASSADOR
Unlock exciting opportunities in clinical research. Join the ABRS team, where innovation, expertise, and collaboration shape the future of healthcare.
Expertise & Capabilities
Copyright © 2026 All rights reserved


