
Advanced Bioresearch Solutions
Clinical research services that improve the identification, activation, training, and management of clinical trials.




1,000 + Ambassadors
250+
Clinical Trials Supported
Watch the video to learn more about
ABRS
Our Impact



































Expertise
Our expertise in monitoring, auditing, training, and site oversight enables us to deliver flexible solutions to biotech and pharmaceutical companies, contract research organizations (CROs), government and public health institutions, academic institutions, and non-profit research sponsors through a regionalized FullSpectrum FSP model across North America, LATAM, Europe, MENA, Central and Southern Africa, and Asia-Pacific.
- about us
Global FSP Partner for Clinical Trial Operations
ABRS is a trusted Functional Service Provider (FSP) offering scalable, regionally aligned support for clinical research. Beyond staffing, we deliver Clinical Oversight Management (COM) to ensure protocol adherence, operational consistency, and full regulatory compliance.
- Know More
We support you
As your strategic partner, ABRS accelerates timelines, safeguards quality, and supports every phase of your trial with adaptable, compliant, and expert-driven solutions.
Our FullSpectrum FSP model is designed to support sponsor-led clinical trial execution, with ABRS acting as a strategic partner while sponsors retain full ownership, decision-making authority, and operational control across all regions.
At ABRS, our dedication to success goes beyond borders, reaching across different parts of the world. With extensive experience in various regions, we offer unmatched expertise in organizing and overseeing clinical trials using our tailored Functional Service Provider (FSP) Model.
- Benefits
Benefits of our Tailored and Scalable FullSpectrum FSP Model
Goals

- Scalable support tailored to evolving study demands resources

Resources

- Cost-efficient resourcing with flexible allocation communications

Communications

- Streamlined decision-making through direct sponsor interaction

Process Stability

- Consistent delivery supported by standardized operational frameworks

Qualified Staff

- Experienced clinical professionals with regional expertise

Project Control

- Sponsor-led oversight with full transparency and governance

- Our Work
Let’s explore
At ABRS, we support clinical development across global regions, combining regional expertise with a deep understanding of local regulatory and operational requirements. Through our tailored FullSpectrum Functional Service Provider (FSP) model, we deliver flexible, scalable support for the organization and oversight of clinical trials worldwide.
Years
Experience and Professionalism
Learn More About Us
At ABRS, our work is guided by three core principles: Patient Safety, Data Integrity, and Adherence to Protocols.These principles shape how we design, manage, and support clinical trials ensuring quality, compliance, and consistency across every region and phase. By embedding these fundamentals into our FullSpectrum service model, we help sponsors execute studies with confidence and control.

- Structured Data Review & Integrity Controls
- Protocol-Aligned Functional Execution
- Audit-Ready Processes & Inspection Support






- Join Us
Submit Request
Submit a request to understand how ABRS can support your goals
We utilize our resources to help YOU
- FAQ
FAQ’s
ABRS offers a comprehensive range of services spanning from study start up to close out, effectively supporting all key phases of clinical research. Our flexible and scalable approach allows us to adapt to the unique demands of each project. As a result, we ensure quality, compliance, and operational efficiency at every step. In other words, we tailor our services to align with your goals while maintaining the highest standards throughout the clinical development process.
ABRS provides critical insight, operational support, and proven best practices to ensure the success of your clinical trials. In addition, we are fully committed to delivering solutions that empower you to innovate with confidence, seize new opportunities, and effectively propel your research forward. Moreover, by combining expertise with flexibility, we help sponsors navigate challenges and accelerate development timelines across all phases of the clinical process. As a result, our partners benefit from enhanced efficiency, reduced risk, and greater strategic alignment. Ultimately, ABRS stands as a dependable ally in driving meaningful outcomes throughout your research journey.
ABRS stands apart by delivering a FullSpectrum FSP model that preserves sponsor control while ensuring governed, high-quality execution. Unlike traditional service models, our role-based approach provides flexible resourcing with full oversight, actionable insights, and consistent execution across all trial phases—enabling informed decision-making without compromising protocol adherence or data integrity.
At ABRS, innovation in clinical research means improving how studies are executed. We apply flexible, role-based FSP model and consistent processes that adapt to each study’s needs while preserving sponsor control. This approach helps sponsors manage complexity efficiently and move trials forward with confidence, while maintaining data integrity, patient safety, and protocol adherence.
With experience supporting clinical trials across regions worldwide, ABRS offers a truly global approach to clinical research. Our teams are equipped to navigate regional regulatory requirements while maintaining consistency and quality, helping sponsors advance studies efficiently across geographies.
ABRS understands the cultural challenges that shape clinical research across different regions. Our work is supported by a global network of clinical research professionals—internally known as Ambassadors—who are carefully selected based on their clinical expertise, regional experience, and cultural competence. These professionals are trained to combine technical rigor with strong interpersonal skills, enabling respectful collaboration, effective communication, and consistent execution across diverse regional environments.
Our Location
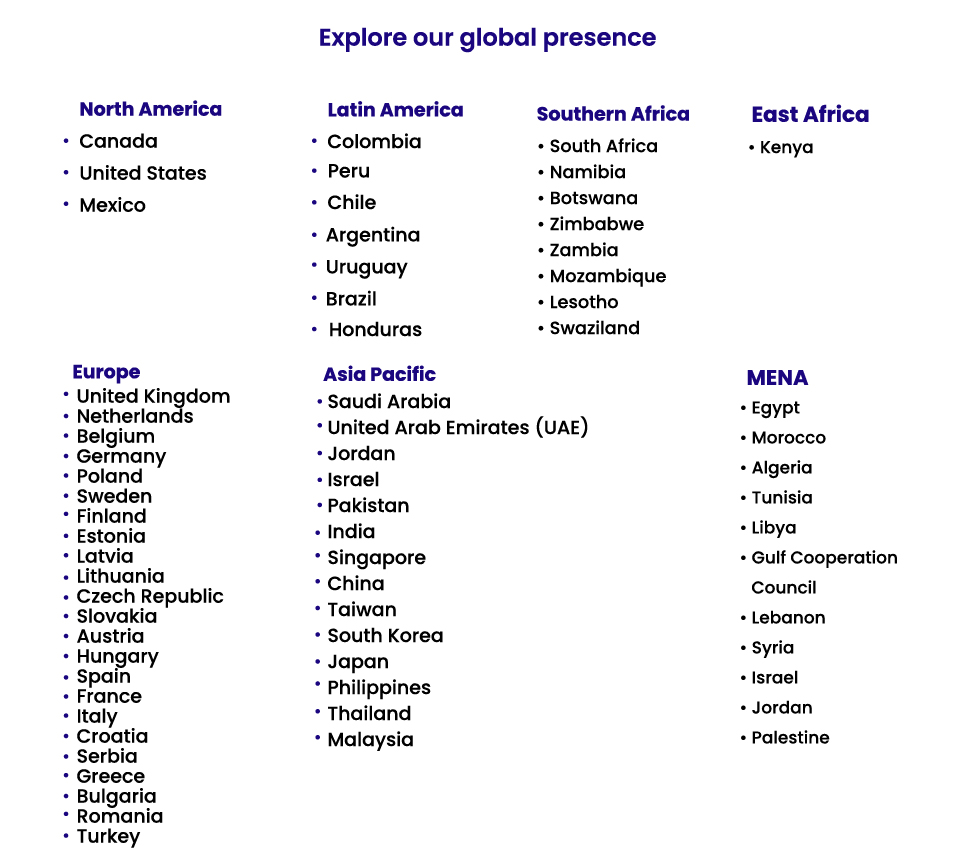
At ABRS, we currently provide coverage
North America
Latin America
East Africa
Southern Africa
Europe
Asia-Pacific
MENA
- Our Blog
Our Latest Articles
Operational Governance in Global Clinical Trials: Bridging Regulatory Expectations and Executional Excellence
By: ABRS- Clinical Insights Team Abstract The modernization of Good Clinical Practice through ICH E6(R3) represents a structural transformation in...
Understanding the Importance of Site Oversight in Clinical Trials
By: ABRS- Clinical Insights Team Abstract As clinical research continues to evolve, increasing protocol complexity, advanced therapies, decentralized study components,...
Why Emerging Regions Are Playing an Increasing Role in Global Clinical Development
By: ABRS- Clinical Insights Team Abstract Global clinical development is undergoing a geographic shift. As sponsors seek more representative data,...
The Three Core Principles of ABRS
Patient Safety
Data Quality
Adherence to protocols

Join us as an ABRS AMBASSADOR
Unlock exciting opportunities in clinical research. Join the ABRS team, where innovation, expertise, and collaboration shape the future of healthcare.
Quick Links
- Blog
- Articles
- Expert Interviews
- Therapeutic Expertise
- Subject Matter Experts
Expertise & Capabilities
- Tailored Functional Service Provider
- Clinical Oversight Management (COM)
- Clinical Research Services North America, LATAM, Europe, MENA, Central and Southern Africa, and Asia-Pacific.
Copyright © 2026 All rights reserved


